Researchers discover a way to stop the spread of HPV
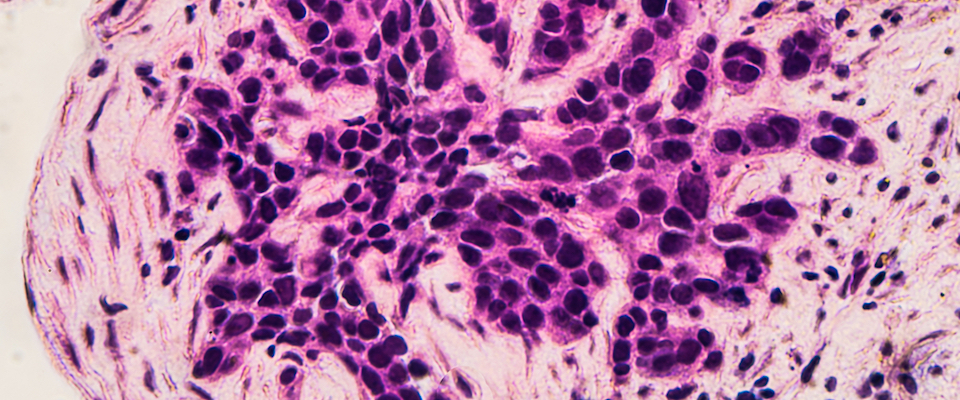
Any woman who’s had less-than-totally-protected sex has spent at least one sleepless night worrying about human papilloma virus, the sexually transmitted disease that causes genital warts and cervical cancer. Though some women’s bodies rid themselves of the virus within a few months, others live permanently with the disease—and its stigma. Eighty percent of women contract the virus by age 50—and many don’t even know it. Worldwide, half a million women are diagnosed with cervical cancer each year, and a quarter million die.
A much-heralded preventive vaccine called Gardasil, approved last year, promises to reduce the number of new infections. And now a Berkeley research team led by Michael Botchan, a molecular biologist at Berkeley’s Institute for Quantitative Biology, is marching toward a cure for those already carrying the virus.
Botchan and re- searchers Eric Abbate and Christian Voitenleitner set out to determine how the virus spreads. With a degree of serendipity and good luck, they also ended up discovering a way to stop that spread.
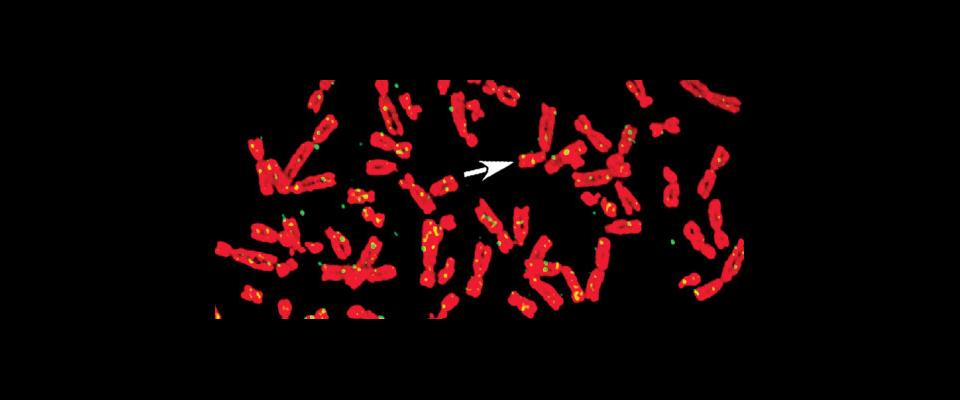
HPV hides out in skin cells; it travels by clinging to chromosomes as one cell divides into two. Without that clinging ability, freefloating virus plasmids would stray into far-off nether regions of the cell, and they’d be left out of new cells’ nuclei. That would render the virus impotent: unable to replicate and unable to survive.
The Berkeley team discovered that HPV clings to human chromosomes the same way BPV—bovine papilloma virus—clings to cow chromosomes: An HPV protein, called E2, gloms onto the tail of a body cell protein, called Brd4. The two proteins fit together much like a key fits into a lock. Meanwhile, the other end of Brd4 attaches itself to a chromosome, and, voila, the wily virus has hitched a ride into a new cell’s nucleus.
After documenting this lockand- key mechanism, bamboozling the virus was a natural next step. The researchers inundated HPVinfected cells with copies of an imitation “key”: a short section of protein, or a peptide, that fits into the virus’s “lock” but does not allow it to attach to chromosomes. It worked: The viral plasmids fastened themselves almost exclusively to fake keys. Gummed up, they failed to bind with host chromosomes.
“They’re locked out of the nucleus,” says Voitenleitner.
The team hopes that bit of trickery will eventually result in a pill or cream that cures HPV. Though that development is still years away, it provides a measure of hope for women already infected with the virus, especially in developing countries, where the incidence of cervical cancer is much higher than in the U.S., and where the HPV vaccine—the most expensive vaccine ever developed—will likely remain largely out of reach.
From the March April 2007 Centennial Edition issue of California.