From diagnostics to warfare, a sampling of how gene-editing is being used to rewrite the world’s DNA.
In October, UC Berkeley professor Jennifer Doudna shared the Nobel Prize in chemistry for the discovery of CRISPR/Cas9, a gene-slicing tool that can be programmed to make precise edits to DNA. Since its discovery, CRISPR has captured the imaginations of everyone from pig farmers to infectious disease researchers. Here are just some of the ways it is already being put to work.
Agriculture
Animal and plant pathogens pose a serious threat to global agriculture. To combat this, Berkeley-based Caribou Biosciences Inc. is exploring ways to use CRISPR to make livestock less susceptible to disease. Scientists at Cal’s Innovative Genomics Institute, meanwhile, are developing gene-editing techniques to make cacao more resistant to disease and drought in the face of rising global temperatures.
Disease Prevention
Berkeley researchers are hoping to use CRISPR to sterilize female mosquitoes to control the spread of malaria, dengue, and other mosquito-borne illnesses. Known as a gene drive, the technology propagates a particular set of genes throughout entire populations, offering a cost-effective and, advocates contend, environmentally friendly solution to insect-borne disease.
Therapeutics
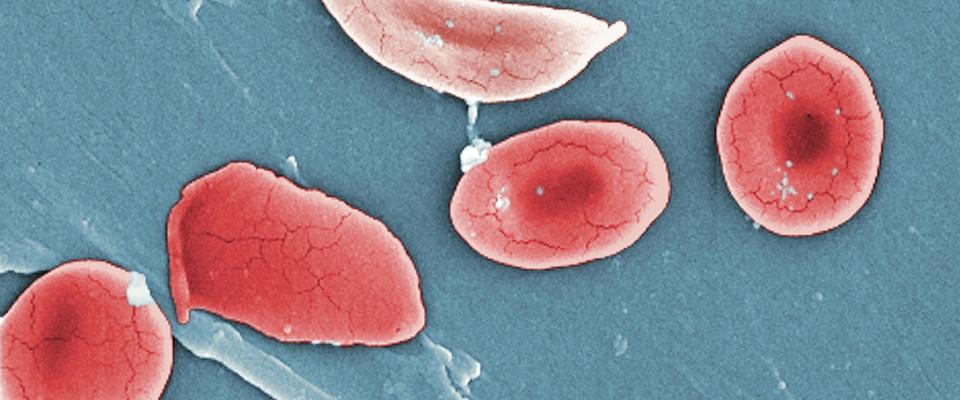
With CRISPR in our toolbox, human genetic disease may become a thing of the past. Researchers from Berkeley, UCSF, and elsewhere have used CRISPR to fix the mutated gene that causes sickle cell anemia. Berkeley bioengineering professors have also used CRISPR to repair the mutation that causes muscular dystrophy in mice. Soon, gene editing might be used to treat viral infections, inflammatory diseases, and even cancer.
Diagnostics
In the age of COVID-19, the need for rapid, reliable diagnostics for infectious diseases is more urgent than ever. Mammoth Biosciences—co-founded by Jennifer Doudna—has developed nucleic acid tests that use CRISPR to detect the virus in less than an hour. Mammoth is also developing CRISPR-based diagnostic tests for non-infectious diseases, including cancer.
Warfare
At Berkeley’s Innovative Genomics Institute, professor Fyodor Urnov is co-leading an initiative to identify genes that, when turned on or off, could protect soldiers from acute radiation sickness. With funding from DARPA, the researchers also hope to develop CRISPR-based medicines to both prevent and treat radiation sickness.
Germline Edits
In 2018, scientist He Jiankui drew criticism for using CRISPR to make germline edits—alterations that are passed to future offspring—in human embryos, despite the potential dangers and taboo against it. While Dr. He insisted he was protecting the babies from HIV, critics fear that the technology could be used for more nefarious ends. For more on the implications of He’s experiment, listen to the fifth episode of California’s podcast, The Edge, “Can You Make Your Baby Glow?”